Interim Nonclinical
Study Monitoring
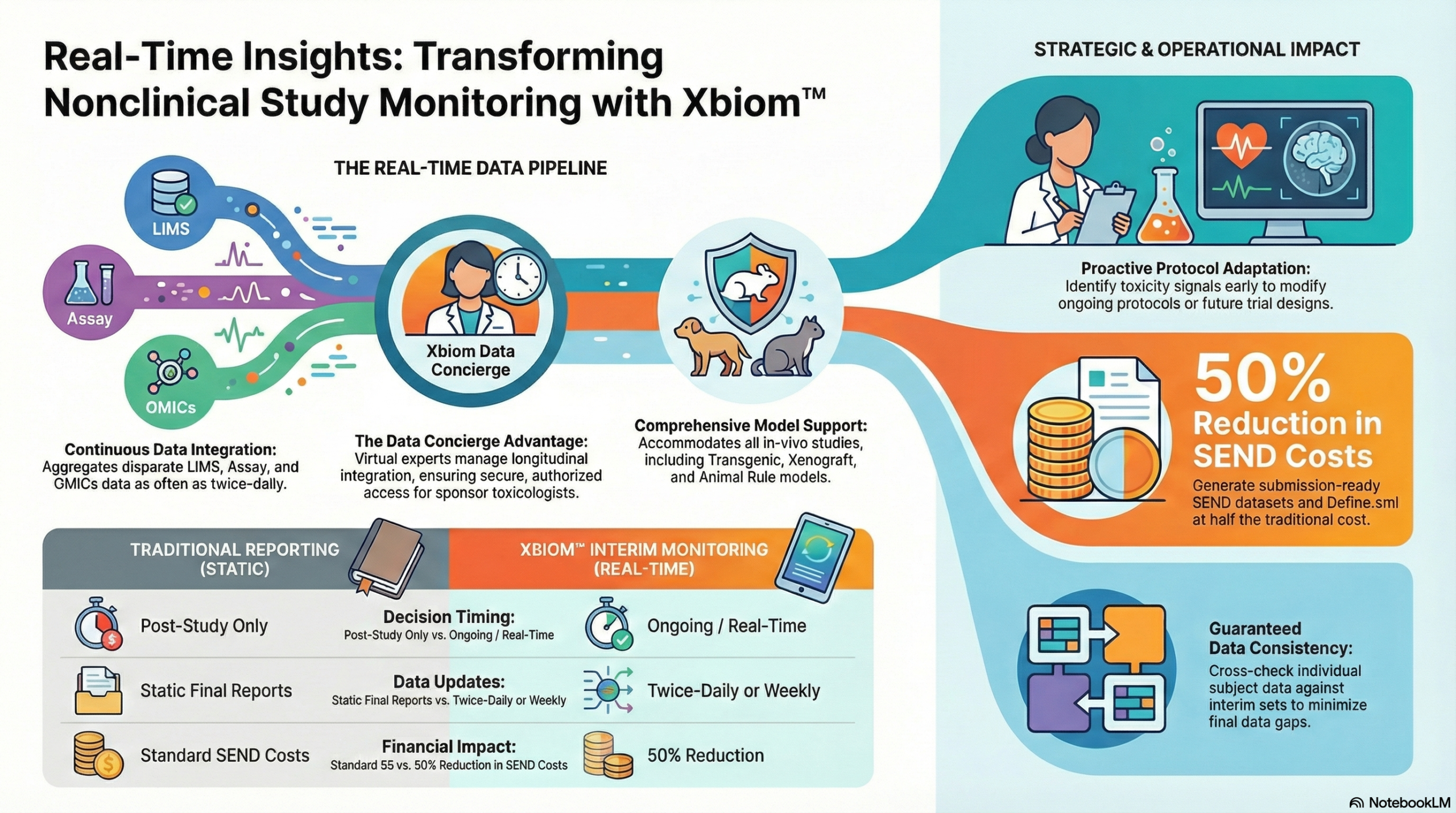
Make data-driven decisions while your study is ongoing. Real-time integration of disparate study data and analytics on Xbiom™ Insights.
Real-Time Data Integration
Study Reports published after completion offer little opportunity for on-study decisions. Xbiom™ enables you to identify toxicity signals early, adapt protocols, and consider new trial designs by monitoring interim data loaded as often as twice-daily.
Our solution aggregates and integrates disparate LIMS and Assay data from multiple sources, making it available to sponsor toxicologists in near-real-time.
Empowered Decisions
The Study Monitor is empowered to decide with agile aggregation of data from multiple sources.
The Xbiom™ Advantage
Delivered on a private, encrypted, cloud-based environment for secure, authorized access.
Data Concierge
A virtual team of data scientists and statisticians collecting and loading cytokines, bioanalytic, OMICs, and other data.
Any Data Source
Integrates disparate LIMS and Assay data. Accommodates all in-vivo studies including Transgenic, Xenograft, or Animal Rule models.
Intuitive Analysis
Analyze data in any way you wish, with any grouping or cohorts, on a powerful and intuitive data platform.
Consistency Check
Final datasets can be cross-checked against the interim set, assuring data consistency and minimal data gaps.
Cost Savings
SEND datasets with nSDRG and Define.xml can be generated from this integrated data at 50% of the indicative SEND cost.
Expert Consults
For small BioTechs, we also offer board certified toxicologists for consults.
Minimize Surprises & Data Gaps
Periodic analysis and visualization while monitoring your study means fewer surprises when the final dataset is prepared.