Monitoring Clinical Trials
with Interim Data
Data driven decisions on ongoing clinical trials. Real-time integration of EDC Patient Data and Biomarker data from Bio-Samples.
Bench-to-Bedside Translational Medicine
Biotechs developing biologic therapeutics based on genomic or immuno-biomarkers benefit from closely monitoring ongoing trials to identify key biomarkers or responders early.
The Challenge: Traditional workflows take weeks to months to convert EDC data to SDTM and ADaM.
The Solution: We electronically read and curate EDC and Biomarker data into a Universal Data Model (UDM) that is inherently longitudinal patient aligned, available within 24 hours.
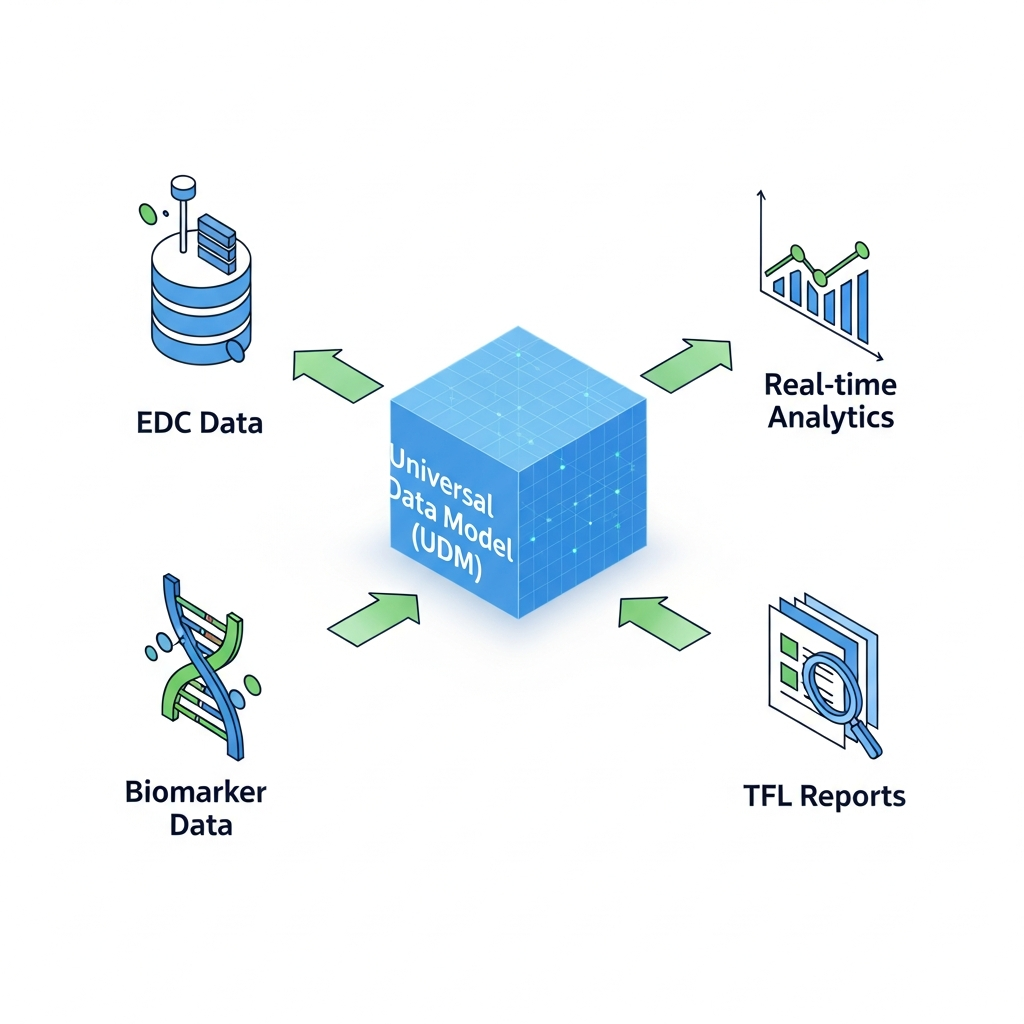
Our Approach
Accelerate scientific research and development through advanced data integration and analytics.
Unified Data Model
Convert EDC and Biomarker data into a longitudinal patient-aligned UDM secured on Xbiom™.
Rapid Availability
Make analysis-ready data available within 24 hours of reading data updates.
Cohort Selection
Search, find, and select cohorts meeting stratified criteria for comparative analysis.
TFL Generation
Capture insights with downloadable TFL objects that auto-update with new data loads.
Collaboration
Search, find, collaborate and publish TFLs into reports and posters.
Sample Identification
Identify patients needing bio-sample assays to fill data gaps or for new panels.
Accelerate Your R&D
Access Xbiom™ insights technologies under a monthly subscription, along with our Data Concierge service—a virtual team of physicians, bioinformatics, and data scientists.