Biomarker Assays &
Omics Data Intelligence
Accelerate clinical insights and reduce attrition rates by validating lead candidates early with automated, standardized biomarker data transformation.
Precision Medicine Requires Precise Data
Biomarker assays serve as critical pharmacodynamic markers for monitoring clinical efficacy and understanding mechanisms of action. Early detection of intermediate success endpoints shortens follow-up times and supports tighter development timelines.
Early Efficacy Signals
Specific biomarker assays can provide preliminary indications of a drug’s effectiveness. Teams can assess response patterns earlier and make informed go or no-go decisions before investing heavily in late-stage trials.
Resource Optimization
Clear assay readouts early in development reduce attrition and help focus budget and operations on candidates with evidence of clinical benefit.
Omics and Immuno-Assays
Support for genomics, transcriptomics, proteomics, metabolomics and immunology data across clinical and nonclinical programs.
Specialty Assays
Omics or immuno-assays, ADA, FACS, IHC, bio-analytics and other specialty assays that complement standard lab pathology for biologics and advanced therapies.
Automated Standardization
Assay outputs are transformed to standard notation using globally accepted registries so datasets remain comparable and analysis ready.
Supported Omics Ontologies
Xbiom maintains awareness of standards in globally accepted registries and applies these definitions when transforming biomarker assay data. This preserves scientific meaning while enabling consistent reporting across studies and vendors.
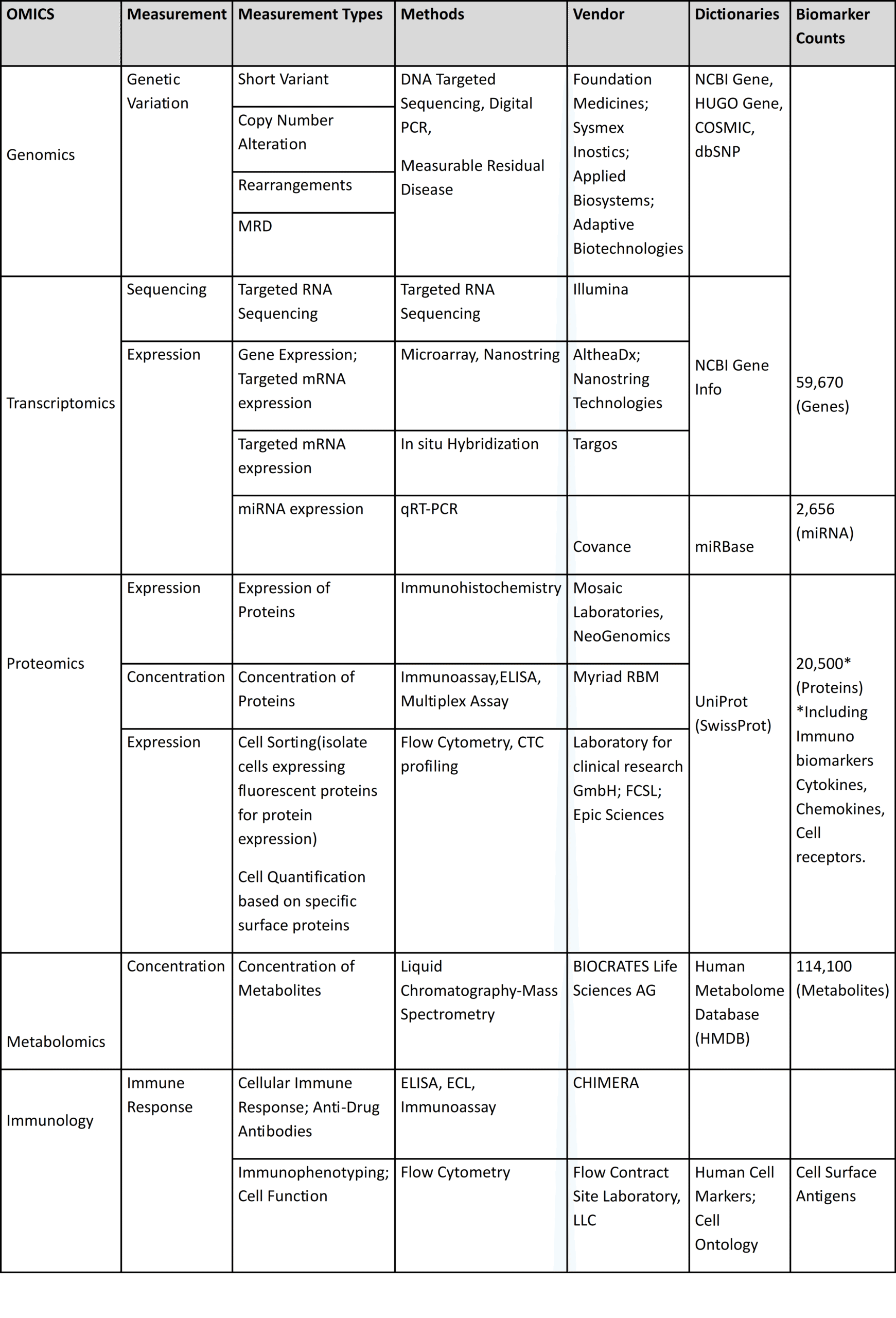
Representative view of Omics categories, measurement types, methods, vendors and dictionaries supported in Xbiom ontologies.
Enterprise Data Intelligence
A single environment to curate, standardize and analyze complex biomarker streams from discovery through regulatory submission.
Standardized Notation
Automated mapping of raw assay outputs to controlled dictionaries, units and code lists suitable for analysis and reporting.
Cross‑Study Integration
Harmonize biomarker data across trials and programs to detect consistent patterns and translational signals.
Regulatory Readiness
Data structures that support SEND and SDTM alignment for biomarkers used in nonclinical and clinical submissions.
Multi‑Omics Support
High‑dimensional datasets from sequencing, microarrays and mass spectrometry managed in a consistent, queryable model.
Vendor‑Agnostic Ingestion
Consolidate outputs from multiple CROs and central labs while preserving provenance and assay context.
Search and Discovery
Locate specific biomarkers, genes or variants across the repository to support translational research questions.
Ready to standardize your biomarker data?
Talk with our data scientists about integrating Omics and specialty assay outputs into a unified, governed asset for clinical and nonclinical programs.