ADaM for
Clinical Analysis
Analysis-ready datasets consistent with Statistical Analysis Plans. Facilitating statistical modeling, regulatory submissions, and reproducible research.
Analysis Data Model (ADaM)
Analysis of clinical trials is a necessary step to assess the efficacy and safety of novel therapeutics. The Analysis Data Model (ADaM) is an exchange standard of CDISC designed to hold as-collected patient data consistent with the Statistical Analysis Plan.
ADaM datasets structure as-collected SDTM data in a form that is analysis-ready, allowing seamless execution of statistical models, efficacy evaluations, and safety analyses.
Traceability
Direct links to SDTM and source data ensuring clear audit trails.
Compliance
Endorsed by FDA and PMDA for regulatory submissions.
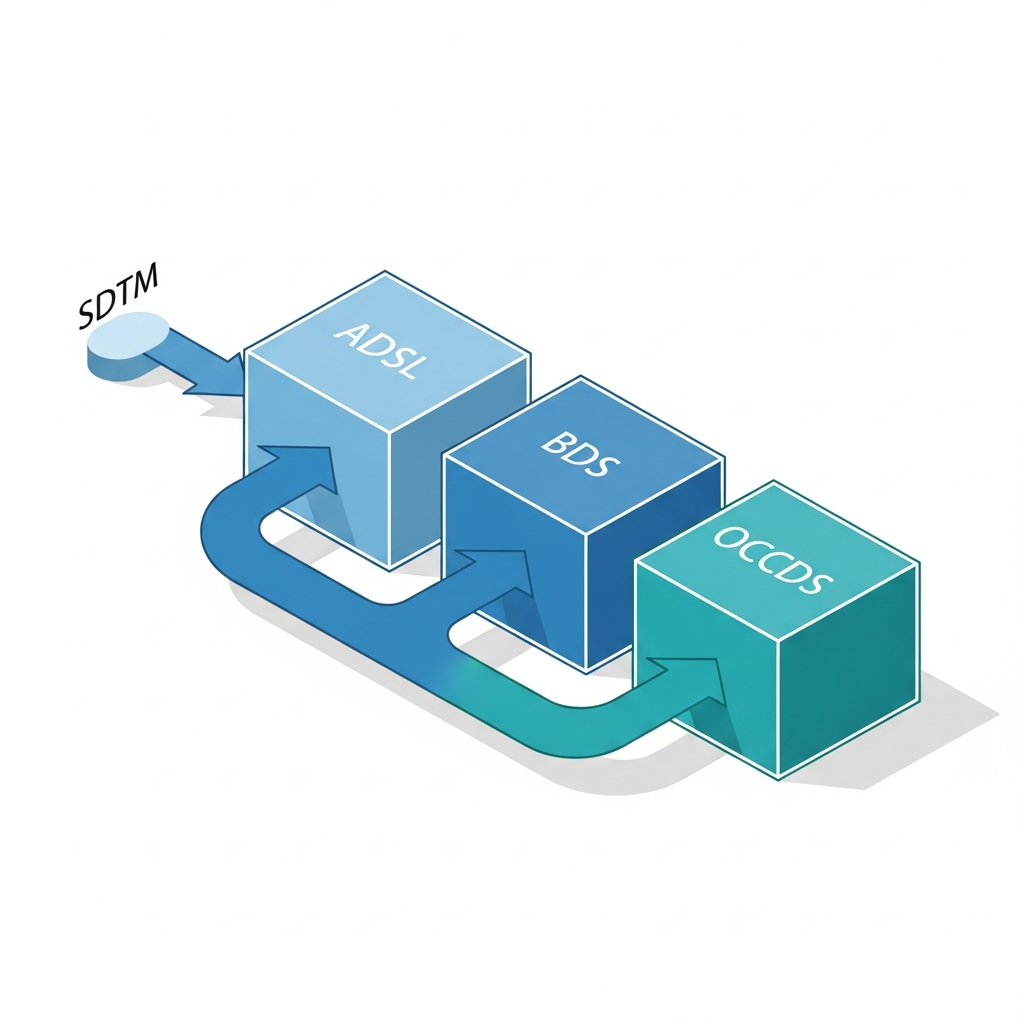
Standardized Data Structures
ADaM provides a standardized framework that includes specific data structures for different analysis needs.
ADSL
Contains demographic and treatment information for each subject.
BDS
Supports longitudinal and time-to-event analyses.
OCCDS
Handles event-based data, such as adverse events.
Xbiom™ MDR & Smart Transformer
Our modules support generating compliant exchange datasets (SDTM & ADaM) that ensure data consistency, reliability, and traceability.
- ✓ Generate ADaM artifacts: define.xml & Reviewer's Guide
- ✓ Automated validation against CDISC/FDA/PMDA rules
- ✓ Data Services Layer for view-ready research data
Regulatory Submission Package
Validation of analysis data for compliance with regulatory standards is an important step in submission pathways.