SEND ASSURE
QC of SEND Datasets
Turnkey quality control for CRO and 3rd party generated SEND datasets. Validated against final PDF Study Reports, metadata, and CDISC conformance rules.
Turnkey QC Service
This service provides a thorough check of CRO and 3rd Party Generated SEND datasets against the final PDF Study Report, its metadata, and tabulated summary data.
Datasets are validated against all applicable conformance rules of CDISC, FDA, PMDA with eDataValidator; and SEND IG best practices.
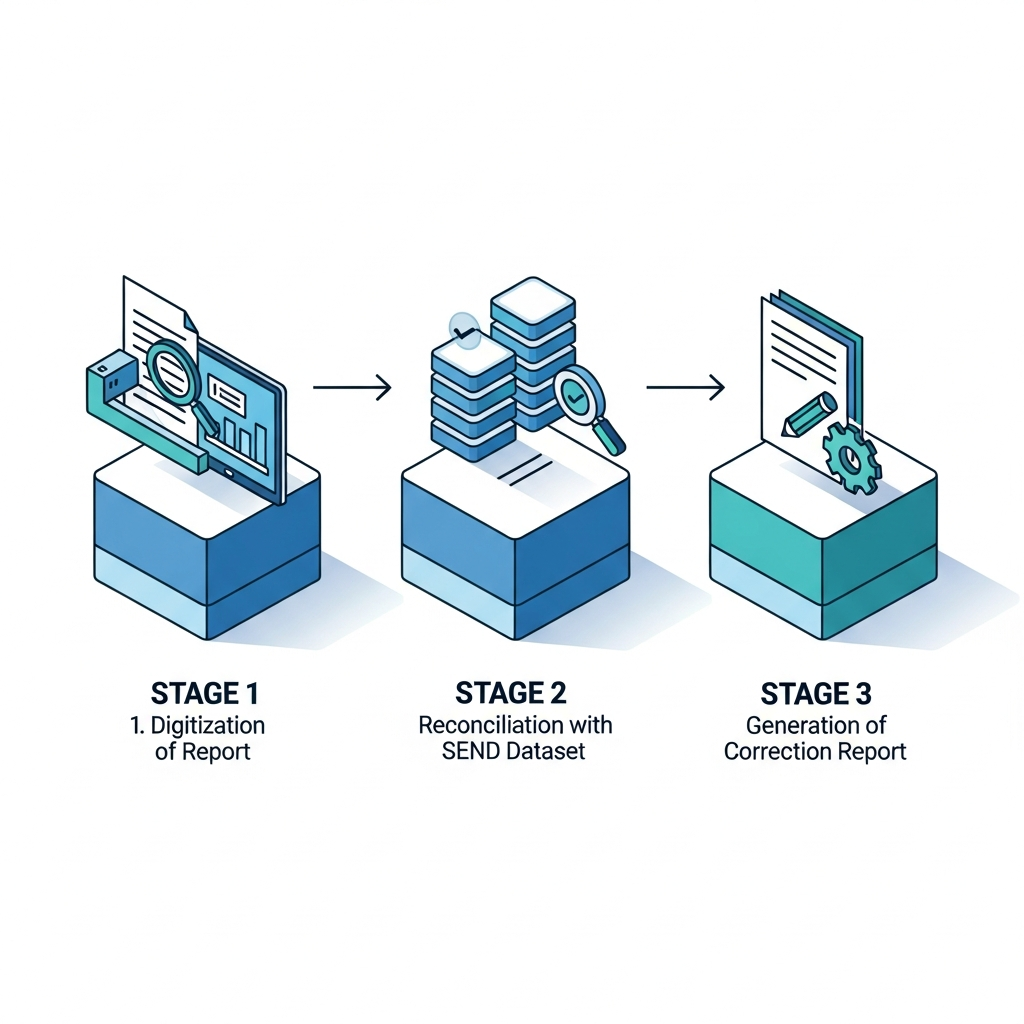
Digitization First Approach
We digitize the Study Report to extract essential metadata and summary tabulations, then reconcile them against the SEND dataset to ensure they match perfectly.
Business Benefits
Why sponsors trust SEND ASSURE for their critical regulatory data.
Study Report Consistency
Ensures consistency with the audited GLP Study Report as the trusted reference.
100% Data Check
We do NOT rely on spot-checks. We check 100% of the subject data by regenerating summaries.
TCG Compliance
Checks that SEND dataset, Define.xml and nSDRG meet TCG rules and avoid TRC conditions.
⚠️ If SEND Preparer Fixes Errors
- ✓ Consolidated Quantitative & Qualitative Comparison Report
- ✓ SEND Assure Report (Excel) with suggested corrections
- ✓ Return of original SEND Dataset and Study Report
💾 If PointCross Fixes Errors
- ✓ Submission Ready SEND Package (xpts, nSDRG, Define.xml)
- ✓ Documentation of Consistency, Conformance and Quality Checks
- ✓ Consolidated Comparison Report with corrections applied